The Bromination takes place at tertiary carbon when the alkane is treated with Br2 in the presence of heat because the tertiary alkyl radical is more stable than secondary and primary alkyl radical.
By the action of heat, Br2 breaks into 2Br radicals as shown below:

There are 8 possible sites for removal of hydrogen by bromine radical as shown below:
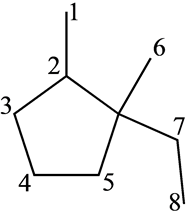
All the labeled sites contain one or more than one hydrogen groups.
The number of hydrogen attached to each site is as follows:
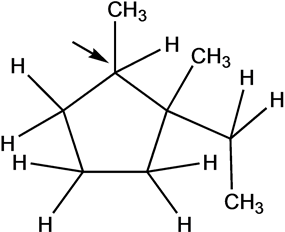
C-1 carbon has less number of hydrogen. Therefore, radical is form at C-1 carbon as shown below:
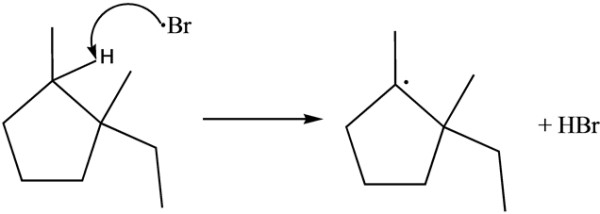
There is one methyl group at C-1 carbon and one methyl and one ethyl group at C-2 carbon of cyclopentane. After the removal of hydrogen, more substituted carbon forms stable radical but there must be hydrogen group present to form radical.
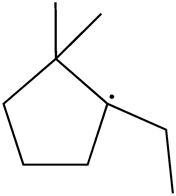
There are three possible shifts; one is methyl shift from C-2 to C-1 carbon that will result in following radical:
The suffix is ane
The IUPAC name of the given structure is 3-ethyl-2,2-dimethylhexane.