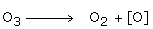1. SO2 -the oxidation state of sulphur is +4, it can loose two more electron to reach upto +6(that exists in its compound).Moreover it can gain more electrons to reach the oxidation state of -2(lowest oxidation state observed in its compound).It means it have capability to loose and gain both so it can work as oxidizing as well as reducing agents.
2. In case of H2O2 , Oxygen is in -1 oxidation state it can loose one electron(will be zero oxidation state as in O2) as well as gain one electron(oxidation state will be-2 as in oxides) hence can work as both oxidizing as well as reducing agent.
3. HNO3- Oxidation state of nitrogen is +5 , This is the maximum oxidation state show by nitrogen in its compound.so it can not loose electron further but it can gain electron hence can act as oxidant only.
4. Ozone acts as a powerful oxidizing agent due to the reaction,