Types of Tautomerism:
1. Keto enol tautomerism
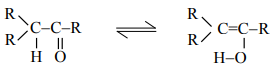
2. Phenol−Keto tautomerism:

In this case, enol form is more stable than keto form because of the aromatic stabilization.
3. Nitroso−Oxime tautomerism:

This equilibrium lies far to the right and as a rule nitroso compounds are stable only when there is no α−hydrogen atom.
4. Nitro−Aci tautomerism:
Aliphatic nitro compounds are in equilibrium with the aci forms.

The nitro form is much more stable than the aci form because nitro group has resonance. Aci form of nitro compounds is also called nitronic acids.
5. Imine−Enamine tautomerism / cyanide−enimine tautomerism:

Imine form predominates generally. Enamines are stable only when there is no hydrogen atom attached to nitrogen.
