Freundlich Adsorption Isotherm:
The variation of extent of adsorption (x/ m) with pressure (P) at a particular temperature was given mathematically by Freundlich in 1909. From the adsorption isotherm, the following observations can be easily made :
(i) At low pressure:
The graph is almost straight line which indicates that (x/ m) is directly proportional to the pressure. This may be expressed as:
\(\frac { x }{ m } \propto P \, or\, \frac { x }{ m } =kp\) … (1)
Where k is a constant.
(ii) At high pressure:
The graph becomes almost horizontal which means that x/ m becomes independent of pressure. This may be expressed by
\(\frac { x }{ m } =constant\)
or
\(\frac { x }{ m } \propto P^{ o }\) ( ∵ Po = 1)
(iii) At medium pressure:
The value of + depends on the power of pressure whose value is between zero to one.

n and k are constant whose value depend on the nature of adsorbate and adsorbent. This relationship was first given by Freundlich. So it is also called Freundlich adsorption isotherm. By taking logarithm of both sides of equation (3).
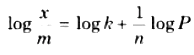
Comparing eq. (4) to y’= c + mx so if graph is plotted between \(log\frac{x}{m}\) and log P, then a straight line is obtained.
The slope of the line will be equal to \(\frac { 1 }{ n } \) and intercept will be equal to log k.
