Fuel cells are the one where chemical energy is converted into electrical energy. The energy can be obtained indefinitely from a fuel cell as long as the outside supply of fuel is maintained. One of the examples is the hydrogen-oxygen fuel cell.
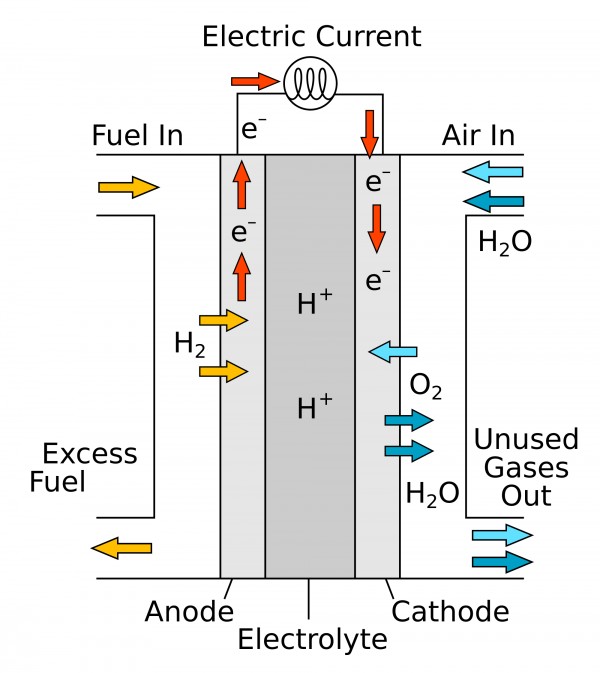
Hydrogen-oxygen fuel cell: The cell consists of three compartments separated from one another by porous electrode. The hydrogen gas is fed into one compartment and the oxygen gas is fed into another compartment. These gases then diffuse slowly through the electrodes and react with an electrolyte that is in the central compartment. The electrodes are made of a conducting material, such as graphite, with a sprinkling of platinum to act as a catalyst, and the electrolyte is an aqueous solution of a base.
The electrodes are made of porous carbon and the electrolyte is a resin containing concentrated aqueous sodium hydroxide solution. Hydrogen is oxidized at anode and oxygen is reduced at the cathode. The overall cell reaction produces water. The reactions which occur are:
Cathode: O2(g) + 2H2O(l) + 4e–-----> 4OH–(aq)
Anode: 2H2 (g) + 4OH–(aq)-----> 4H2O(l) + 4e–
Overall reaction is:
2H2(g) + O2(g) ----->2H2O(l )
Advantages of Hydrogen-oxygen fuel cell: (i) Fuel cells are efficient and free from pollution.
(ii) The only product in the reaction of fuel cell is water which can be removed and the astronauts of a spacecraft can drink it.