Whenever a reaction between an oxidising agent and a reducing agent is carried out, a compound of lower oxidation state is formed if the reducing agent is in excess and a compound of higher oxidation state is formed if the oxidising agent is in excess. This can be illustrated as follows:
(i) P4 and F2 are reducing and oxidising agents respectively.
If an excess of P4 is treated with F2, then PF3 will be produced, wherein the oxidation number (O.N.) of P is +3.

However, if P4 is treated with an excess of F2, then PF5 will be produced, wherein the O.N. of P is +5.

(ii) K acts as a reducing agent, whereas O2 is an oxidising agent. If an excess of K reacts with O2, then K2O will be formed, wherein the O.N. of O is –2.

However, if K reacts with an excess of O2, then K2O2 will be formed, wherein the O.N. of O is –1.

(iii) C is a reducing agent, while O2 acts as an oxidising agent. If an excess of C is burnt in the presence of insufficient amount of O2, then CO will be produced, wherein the O.N. of C is +2.
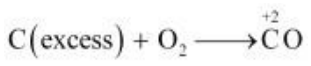
On the other hand, if C is burnt in an excess of O2, then CO2 will be produced, wherein the O.N. of C is +4.
