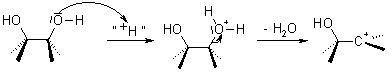
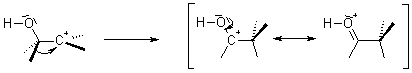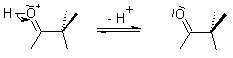Pinacol Rearrangement is also called as Pinacol-Pinacolone rearrangement. It is an acid catalyzed organic chemical reaction in which 1, 2-diols are converted to carbonyl compounds. The name comes from the reactant and the product that is pinacol to pinacolone.
Pinacol Rearrangement
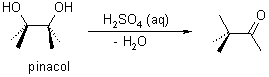
In the conversion that gave its name to this reaction, the acid-catalyzed elimination of water from pinacol gives t-butyl methyl ketone.
Mechanism of the Pinacol Rearrangement
This reaction occurs with a variety of fully substituted 1,2-diols, and can be understood to involve the formation of a carbenium ion intermediate that subsequently undergoes a rearrangement. The first generated intermediate, an α-hydroxycarbenium ion, rearranges through a 1,2-alkyl shift to produce the carbonyl compound. If two of the substituents form a ring, the Pinacol Rearrangement can constitute a ring-expansion or ring-contraction reaction.